New Medicare Part D Demonstration to Address Proposed Changes to Anti-Kickback Safe Harbors
Highlights
HIGHLIGHTS:
- The Centers for Medicare & Medicaid Services (CMS) has announced a new Medicare Part D demonstration to address the potential implementation of a recent proposed rule that would exclude from anti-kickback safe harbor protection pharmaceutical manufacturer rebates paid to Part D sponsors and their contracted pharmacy benefit managers.
- If this proposed rule were finalized to take effect during the 2020 plan year, CMS would then allow Part D sponsors to apply for and participate in the voluntary, two-year demonstration.
- The announcement is a clear acknowledgement by CMS that the proposed changes to the anti-kickback safe harbors could significantly impact the Part D program.
The Centers for Medicare & Medicaid Services (CMS) announced on April 5, 2019, a new Medicare Part D demonstration to address the potential implementation of the recent proposed rule issued by the U.S. Department of Health and Human Services' Office of Inspector General (OIG) that would exclude from anti-kickback safe harbor protection pharmaceutical manufacturer rebates paid to Part D sponsors, Medicaid managed care organizations and their contracted pharmacy benefit managers (the Proposed Rule).1 If OIG were to finalize the rule to take effect during the 2020 plan year, CMS would then allow Part D sponsors to apply for and participate in the voluntary, two-year demonstration.
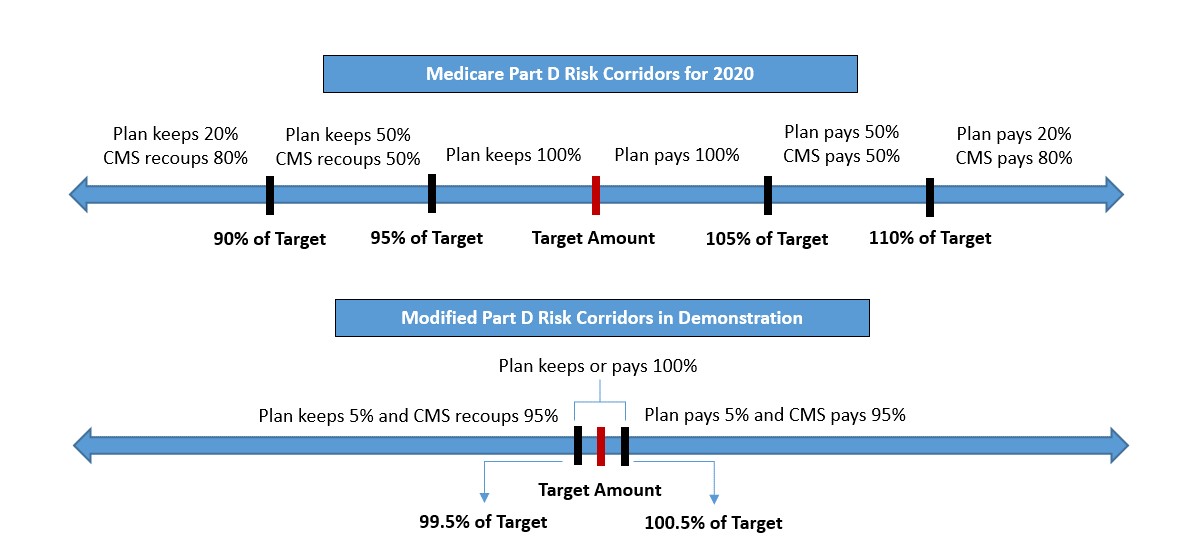
Although CMS is still developing further guidance on the model and application process, the demonstration is constructed to narrow the Part D risk corridors and allow the government to assume a greater degree of risk sharing than current thresholds now permit. The demonstration would modify the Part D risk corridors so that once end-of-the-year costs are reconciled, "the government would bear or retain 95% of the deviation between the target amount, as defined in section 1860D-15(e)(3)(B) of the Social Security Act (the Act) and the actual incurred costs, as defined in section 1860D-15(e)(1) of the Act, beyond the first 0.5%."2
CMS Industry Call Highlights
CMS provided further insight on the planned demonstration during its hosted industry call on April 8, 2019. Please see below for a summary of key takeaways:
- Bid Submission for 2020. Bids must be submitted by Part D sponsors for the 2020 plan year by June 3, 2019. Given that comments on the Proposed Rule were due on April 8, 2019, and that any finalized rule of this significance would require at least another 60 days after publication for implementation, CMS expressly confirmed that Part D sponsors should submit their 2020 plan year bids based on the existing anti-kickback statute and safe harbors. Notably, CMS indicated that it would not allow for submitted bids to be readjusted to accommodate finalization of the Proposed Rule for the 2020 plan year, and that sponsors would continue to be paid their monthly capitated rates as determined. However, participation in the demonstration would allow sponsors retroactively to share a great deal more of the downside risk with the government.
- Eligibility for Participation. Participation in the demonstration would be limited to those Part D sponsors submitting bids. This would exclude any Part D employer group waiver plans (EGWP) from applying for and participating in this model. Part D sponsors may apply for the demonstration at the plan benefit package (PBP) level, which means that CMS would not require sponsors to submit for participation all Part D plans offered by the organization. CMS also confirmed that participation in the demonstration must be for the full two years.
- Implementation of Demonstration. CMS would initiate this demonstration only if the Proposed Rule becomes final and effective during the 2020 plan year. If the Proposed Rule were to be finalized but effective only for the 2021 plan year, as an example, the demonstration would not proceed as planned for 2020. CMS confirmed that the application process to participate in the demonstration would be released soon after a decision is made by the OIG to finalize the Proposed Rule, and no later than the end of this calendar year in order to allow for applications to be submitted in time for the 2020 plan year. CMS expects only "limited operational complexity" for Part D sponsors opting to participate in the demonstration, and believes that CMS will bear the brunt of the operational changes necessary to accommodate how risk corridors will be calculated.
- No Formulary Changes. CMS confirmed on the industry call that it would not reopen the formulary submission window to accommodate new or revised pharmaceutical manufacturer discount structures or arrangements that may be in play with the finalization of the Proposed Rule. The demonstration instead would be strictly limited to the implementation of the modified Part D risk corridors.
Conclusion and Considerations
CMS emphasized that its announcement of the demonstration should not be interpreted as a sign of things to come but rather is intended to provide Part D sponsors with as much notice as possible of alternatives that would be available, should the Proposed Rule be finalized, and to "smooth" the transition for sponsors and beneficiaries. The announcement is clearly an acknowledgement by CMS that the proposed changes to the anti-kickback safe harbors could significantly impact the Part D program. CMS urged sponsors to make their own decisions to participate in the demonstration based on each plan's assessment of its rebate arrangements and the anticipated risks involved if the safe harbor changes are implemented. Holland & Knight's Healthcare & Life Sciences Team will continue to monitor any further guidance from CMS or the OIG on these issues.
Notes
1 84 Fed. Reg. 2340 (Feb. 6, 2019).
2 "Guidance Regarding Part D Bids," CMS Memo from Seema Verma to Part D Plan Sponsors, April 5, 2019.
Information contained in this alert is for the general education and knowledge of our readers. It is not designed to be, and should not be used as, the sole source of information when analyzing and resolving a legal problem. Moreover, the laws of each jurisdiction are different and are constantly changing. If you have specific questions regarding a particular fact situation, we urge you to consult competent legal counsel.
